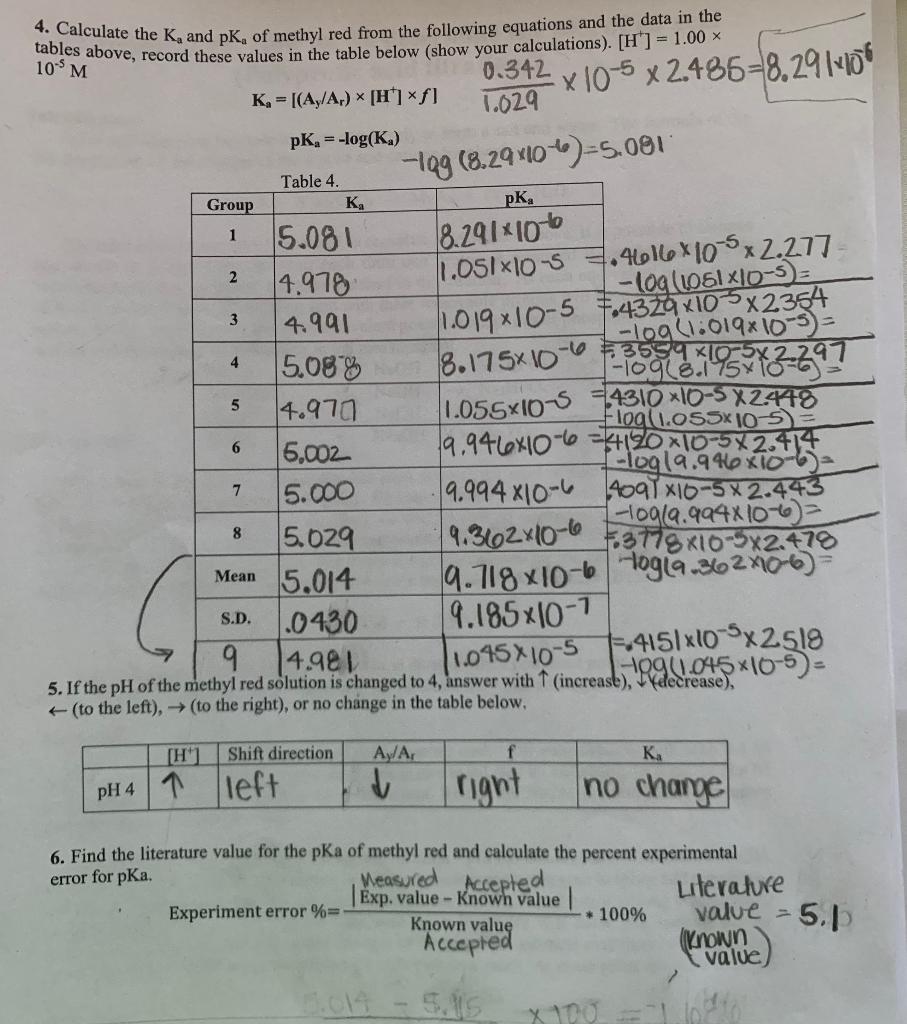
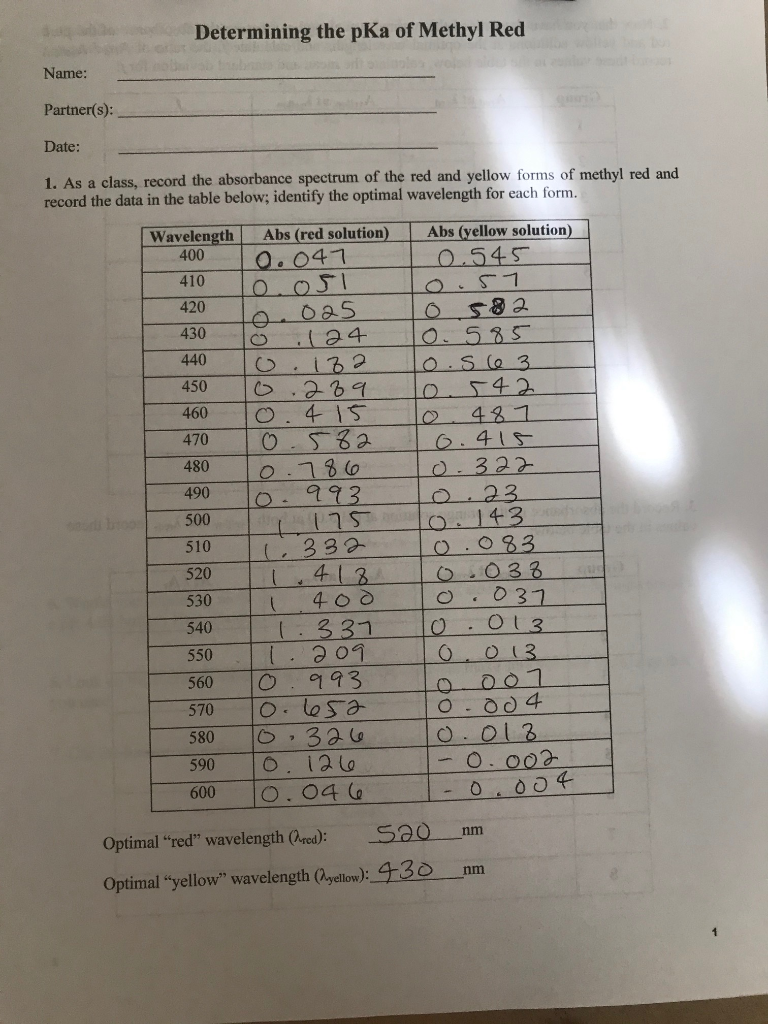
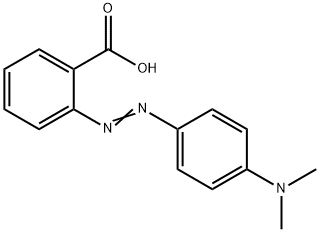
Determination of the Ka of methyl Red Indicator by Colorimetry - Spectroscopy applications: Determination of the Ka of Methyl Red Indicator by | Course Hero

Table 3 from Natural Plant Extracts as Acid-Base Indicator and Determination of Their pKa Value | Semantic Scholar

Determination of the Ka of methyl Red Indicator by Colorimetry Discussion - Spectroscopy applications: Determination of the Ka of Methyl Red Indicator | Course Hero
![PDF] Natural Plant Extracts as Acid-Base Indicator and Determination of Their pKa Value | Semantic Scholar PDF] Natural Plant Extracts as Acid-Base Indicator and Determination of Their pKa Value | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/5760a8648c58e5c7d5b88a0e6de511d2149e292c/5-Table4-1.png)
PDF] Natural Plant Extracts as Acid-Base Indicator and Determination of Their pKa Value | Semantic Scholar

PDF) Determination of Acid Dissociation Constant of Methyl Red by Multi-Peaks Gaussian Fitting Method Based on UV-Visible Absorption Spectrum
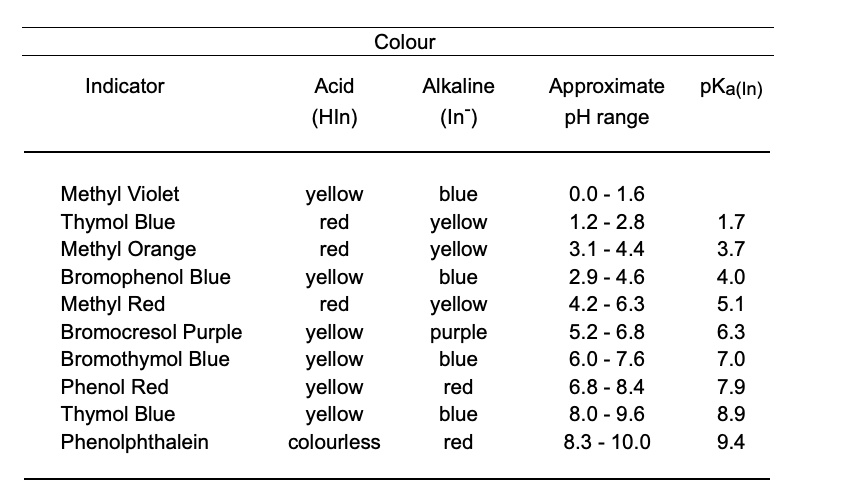
SOLVED: Colour Indicator Acid (HIn) Alkaline (In" ) Approximate pH range pKa(ln) Methyl Violet Thymol Blue Methyl Orange Bromophenol Blue Methyl Red Bromocresol Purple Bromothymol Blue Phenol Red Thymol Blue Phenolphthalein yellow

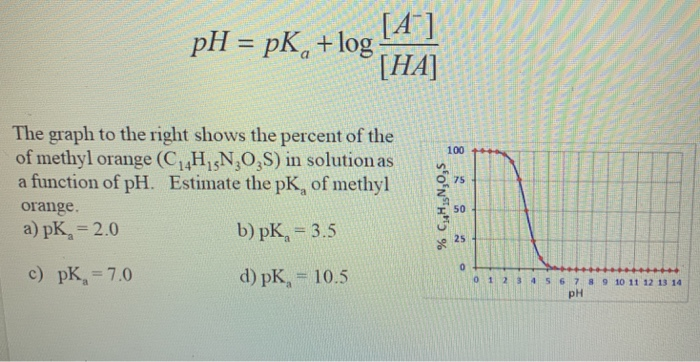
Methyl red has the following structure: It undergoes a color change from red to yellow as a solution gets more basic. Calculate an approximate pH range for which methyl red is useful.

Structures of diazo pH indicators. (A) Main structure of methyl orange... | Download Scientific Diagram
![SOLVED: Calculate the Ka and pKa of methyl red from the following equations and the data in the tables above; record these values in the table below (show your calculations). [H ] = SOLVED: Calculate the Ka and pKa of methyl red from the following equations and the data in the tables above; record these values in the table below (show your calculations). [H ] =](https://cdn.numerade.com/ask_images/a0a70d476fd049fea11245c72f687323.jpg)
SOLVED: Calculate the Ka and pKa of methyl red from the following equations and the data in the tables above; record these values in the table below (show your calculations). [H ] =